Computer System Validation (CSV) Audit Services
Our Computer System Validation (CSV) audit services support biotech and pharmaceutical organizations in establishing, maintaining, and demonstrating compliant computerized systems across the system lifecycle. We partner with quality, regulatory, and IT teams to assess validation governance, data integrity, compliance and inspection readiness for FDA, EMA, and other global health authority expectations.
What is a CSV audit?
A CSV audit is a systematic, independent assessment of computerized systems used in GxP-regulated activities to confirm they are validated, fit for intended use, and compliant with applicable regulatory requirements. This includes alignment with 21 CFR Part 11, EU Annex 11, FDA guidance on computerized systems, and global data integrity expectations.
CSV audits evaluate whether validation activities are appropriately planned, executed, documented, and maintained throughout the system lifecycle, from supplier selection and implementation through change control and system retirement.
Why CSV audits are important:
- Regulatory findings related to inadequate validation or data integrity failures can result in inspection observations or enforcement actions
- Weak CSV governance increases risk to product quality, patient safety, and business continuity
- Poorly controlled computerized systems create inefficiencies, remediation burden, and inspection delays

Our CSV audit services
We provide comprehensive, risk-based CSV audit services covering the following areas:
Software suppliers
Assessment of software vendors supporting GxP activities, including:
- Supplier qualification and ongoing oversight
- Validation documentation and lifecycle support
- Configuration management, release controls, and change management
Electronic records and electronic signatures
Evaluation of systems subject to 21 CFR Part 11 and EU Annex 11, including:
- Electronic record integrity and audit trail functionality
- User access controls and security models
- Electronic signature implementation and governance
Infrastructure and IT service providers
Audits of hosted, cloud-based, and managed service environments, including:
- Infrastructure qualification and responsibility models
- Data hosting, backup, and disaster recovery controls
- Oversight of third-party IT service providers
Statistics and programming environments
Review of validated statistical and programming environments used for GxP activities, including:
- Control of validated tools and libraries
- Change control and version management
- Traceability between requirements, code, and outputs
GxP-related systems
Assessment of computerized systems supporting regulated processes, such as:
- eQuality management systems (eQMS)
- Manufacturing execution systems (MES), Laboratory information management systems (LIMS)
- Clinical, safety, and regulatory platforms
Internal CSV processes
Evaluation of organizational CSV frameworks, including:
- Validation policies, procedures, and standards
- Risk-based validation approaches
- Roles, responsibilities, and governance models

Our Computer System Validation auditing services include:
How our CSV audit process works
Step 1 - Scoping and needs assessment
We align with your organization to define audit scope based on system criticality, regulatory exposure, lifecycle stage, and inspection priorities.
Step 2 - Auditor assignment
A CSV auditor with relevant system, technology, and regulatory experience is assigned based on your environment and compliance needs.
Step 3 - Audit preparation
The auditor develops an audit plan, agenda, and documentation request list, and coordinates with system owners, IT teams, and suppliers as needed.
Step 4 - Audit execution
Audits are conducted remotely or onsite and include interviews, documentation review, and assessment of system controls, validation evidence, and data integrity practices.
Step 5 - Audit reporting
You receive an inspection-ready audit report with a clear compliance narrative, structured observations, and risk-based findings aligned to regulatory expectations.
Step 6 - Follow-up and ongoing support
We support CAPA development, remediation planning, re-audits, and inspection readiness activities to ensure findings are effectively addressed and sustained.
Trusted by top-tier teams worldwide

















650+
Audits conducted in the last year
800
Network of expert GxP auditors
110
Countries covered
Why choose Apotech for CSV audit services?
At Apotech, our onsite and remote CSV audits support pharmaceutical and biotechnology companies in maintaining compliant validated systems and meeting evolving regulatory expectations for computerized systems and data integrity.
Our approach is pragmatic, risk-focused, and grounded in real-world CSV practice across regulated environments.
We differentiate ourselves through:

Global GxP specialists
Experienced CSV auditors with global inspection exposure across FDA, EMA, and other health authorities.

Regulatory and technical expertise
Deep understanding of validation lifecycles, data integrity, 21 CFR Part 11, EU Annex 11, and modern IT and cloud-based environments.

We provide competitive pricing.
Our pricing model is competitive, guaranteeing professional representation that you can trust with no hidden costs.

We speak your language.
Wherever you are in the world, our consultants operate in a wide range of countries and can communicate with you in your local language.
Our Global Reach
We take pride in our extensive global audit network and are committed to utilising local auditors based in-country.
This approach offers several key benefits: local auditors possess in-depth knowledge of regional regulations, communicate effectively in the local language, and help reduce travel expenses for our clients. We have successfully provided support in all the regions highlighted on the map below.
Featured case studies
Medical device CSV
Medical device CSV Introduction IVD manufacturer The client is an IVD manufacturer based in Europe. Although already ISO 13485:2016 certified, they were agnostic when it came to GAMP 5 and 21 CFR PART 11. They have 80 employees based over 2 sites in France. Apotech consultant: Apotech’s consultant coordinated 5 technicians, acting as project manager…
FAQs on Audits
We provide a modern approach to auditing that offers full transparency. We aim to make the process as simple to understand as possible and are always on hand to answer any questions you might have.
Here are some of the questions we get asked most often.
Compliance, standards, and quality assurance
Our CSV audits are conducted in line with applicable regulatory and industry expectations, including FDA guidance, EU Annex 11, ICH principles, and global data integrity standards. All audits are performed independently, with strict confidentiality and secure handling of client information.
Data protection and confidentiality
We apply robust data protection controls and confidentiality practices appropriate for regulated environments and sensitive system data.
Contact us
Whether you need a full CSV audit programme or targeted support for a specific system, vendor, or inspection, we work with you to assess risk, address gaps, and support inspection readiness.
To learn more about our CSV audit and wider quality assurance services, please get in touch today.
Helpful resources
10 CAPA Metrics and KPIs Every Quality Team Should Track
By Imane Nohair, GxP Audit manager at Apotech Corrective and Preventive Actions (CAPAs) are a fundamental element of quality management across the pharmaceutical & biotechnology industries. When designed and executed effectively, CAPAs do more than address isolated non-conformities. CAPA processes strengthen quality systems, reduce regulatory risk, and support sustained compliance across global regulatory frameworks. Across…
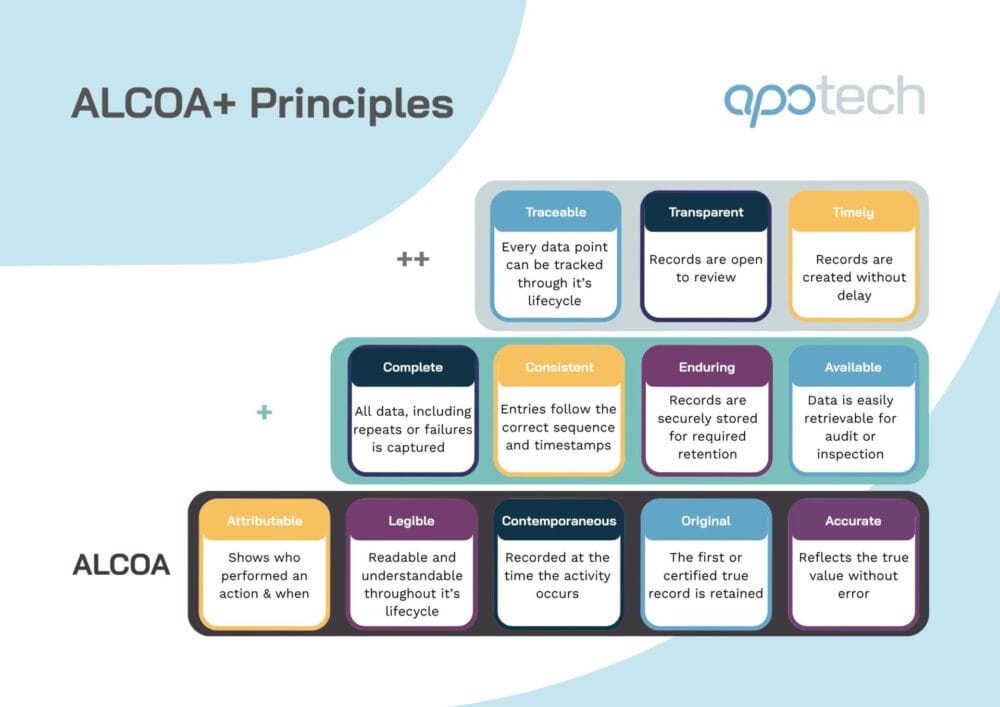
ALCOA+ Principles in Pharma: The Next-Gen Standard for Data Integrity
ALCOA+ and the modern data integrity landscape Data integrity underpins regulatory compliance and patient safety. And as pharmaceutical and biotech organizations adopt increasingly digital systems, automated workflows, and globalized supply chains, regulators including the FDA, EMA, and MHRA have intensified their focus on how data is generated, captured, and maintained. The ALCOA+ principles form the…
GxP Compliance Explained: Beyond Best Practice
Understanding the meaning of GxP is essential for any organization operating in the life sciences sector. In simple terms, GxP refers to “Good Practice” quality and regulatory guidelines, used to ensure products are safe, effective, and reliable throughout their lifecycle. These standards apply across pharmaceuticals, biotechnology, medical devices, and laboratory environments, and are enforced by…