Good Clinical Practice (GCP) Audit Services
Good Clinical Practice (GCP) audits are critical to ensuring clinical trials are conducted ethically, safely, and in full compliance with global regulatory expectations. Our GCP audit services support pharmaceutical and biotech companies at every stage of the clinical lifecycle, helping you maintain compliance, mitigate risk, and prepare confidently for regulatory inspection.
What is a GCP audit?
A GCP audit is a systematic, independent evaluation of clinical trial processes, systems, and documentation against Good Clinical Practice guidelines and applicable regulatory requirements. GCP audits assess whether clinical trials are conducted in accordance with ICH GCP, protocol requirements, and FDA, EMA, and MHRA expectations.
Well-executed GCP audits provide assurance that patient safety, data integrity, and regulatory compliance are embedded throughout your clinical operations. They also play a vital role in identifying gaps early, reducing inspection risk, and supporting inspection readiness across sponsors, CROs, sites, and vendors.
Why GCP audits matter
Without robust GCP audit programmes, organisations face significant risk, including:
- Regulatory findings during FDA, EMA, or MHRA inspections
- Compromised data integrity or patient safety
- Delays to clinical development timelines
- Increased remediation costs and operational disruption
- Reputational damage and loss of stakeholder confidence

Our GCP audit services offering
Our GCP audit services support compliance, oversight, and inspection readiness across the full clinical trial lifecycle. Audits are delivered using a risk-based approach aligned with ICH GCP and global regulatory expectations.
Clinical oversight and third-party audits
Independent audits of external partners involved in trial conduct focused on sponsor oversight, patient safety, and data integrity.
- Investigator site GCP audits
- Clinical Research Organization (CRO) audits
- Vendor qualification and requalification audits
Systems, processes, and documentation audits
Audits of internal systems and processes that underpin compliant clinical trial execution and inspection readiness.
- Clinical systems and internal process audits
- Controlled documentation and SOP reviews
- Trial Master File (TMF) and eTMF audits
Data-focused GCP audits
Specialist audits assessing the integrity, traceability, and regulatory compliance of clinical data and analysis activities.
- Data management GCP audits
- Biostatistics and statistical programming audits
Mock inspections and inspection readiness
Inspection-focused audits are designed to prepare organizations and teams for interactions with health authorities.
- FDA-style mock inspections
- Inspection readiness gap assessments
- Interview preparation and inspection support
How our GCP audit process works
Our GCP audit services follow a structured, risk-based methodology aligned with global regulatory best practice.
Step 1 - Scoping and needs assessment
We define audit scope based on trial phase, regulatory focus, and the clinical oversight model (sponsor, CRO, site, or vendor).
Step 2 - Auditor assignment
A GCP auditor is assigned with relevant clinical trial, therapeutic area, and regulatory inspection experience.
Step 3 - Audit preparation
An audit plan & agenda is developed, including trial documentation, TMF, data, and system considerations.
Step 4 - Audit execution
Audits are conducted onsite or remotely and focus on trial conduct, vendor performance, data integrity, and patient safety.
Step 5 - Audit reporting
Inspection-ready reports provide a clear narrative of what was discovered during the audit, followed by a structured findings and observations section that highlights GCP-relevant risk and the applicable GCP references. Reports can be delivered using either the client’s templates or Apotech’s standard formats.
Step 6 - Follow-up and ongoing support
Following report delivery, we remain available to support CAPA review, clarification with sponsors or vendors, and any required CAPA follow-up activity to ensure findings are understood, addressed, and effectively resolved.

Trusted by top-tier teams worldwide

















650+
Audits conducted in the last year
800
Network of expert GxP Auditors
110
Countries covered
Why choose Apotech for GCP audits?
At Apotech, our onsite or remote audits help pharmaceutical, biotechnology and medical device companies maintain the highest standards of quality and safety. Thanks to our in-house auditing experience, we provide tailor-made solutions that you can trust.

Global GxP Specialists
Our network of 800+ senior auditors covers all GxP disciplines worldwide, providing tailored guidance with local-language fluency

Regulatory and technical expertise
Our auditors bring real-world experience in biotech, pharmaceutical development, and CROs, paired with up-to-date knowledge of ICH, FDA, EMA, MHRA, and other global guidelines. With a minimum of 20 years of experience, they have individually led over 100 audits for various companies across the life sciences sector

We provide competitive pricing
Our local auditors help minimise travel and expense costs, keeping our services cost-effective

Flexible, client-centred service
We offer scalable, tailored GxP audit solutions, from single vendor audits to fully outsourced global programs, adapting to your timeline, scope, and budget. Our dedicated Business and Project Manager model ensures seamless communication, scheduling, and consultant alignment
Our Global Reach
We take pride in our extensive global audit network and are committed to utilising local auditors based in-country.
This approach offers several key benefits: local auditors possess in-depth knowledge of regional regulations, communicate effectively in the local language, and help reduce travel expenses for our clients. We have successfully provided support in all the regions highlighted on the map below.
Featured case studies
GCP Audits – Europe
Seagen is a global biotechnology company dedicated to revolutionizing cancer care. They are Headquartered in the US & Switzerland with a large footprint in the EU.
GxP Audits – Worldwide
GxP Audits – Worldwide Introduction Global Biopharmaceutical Our client is a global biotechnology company dedicated to novel treatments for patients through inhibition of protein kinases to fight cancer. They are Headquartered in the US, with a large footprint in the EU. Types of Audits GCP – For Cause / Investigator / Phase 1 unit GCLP – Central…
GCP Mock inspections – Europe
GCP Mock inspections Europe Introduction Danish R&D Pharmaceutical Company Our client is a pharmaceutical company committed to ensuring compliance with global regulatory standards, particularly FDA requirements for clinical trials Apotech auditors Our Auditors have 20+ years’ experience, all based locally with extensive experience in FDA inspections and GCP compliance. Collectively conducted over 50 regulatory inspections…
FAQs on Audits
We provide a modern approach to auditing that offers full transparency. We aim to make the process as simple to understand as possible and are always on hand to answer any questions you might have.
Here are some of the questions we get asked most often.
Yes, our team can provide comprehensive CAPA (Corrective and Preventive Action) Management support.
Our modern and flexible approach ensures effective handling of CAPA processes, helping not only address and prevent any non-conformities but also contribute towards the continual improvement of both quality and compliance.
Compliance, standards and quality assurance
Apotech operates in alignment with ICH , ISO standards, GxP requirements, and expectations from global regulators including FDA, EMA, and MHRA. All engagements maintain independence, objectivity, and confidentiality.
Data protection and confidentiality
We apply strict data protection measures, GDPR-aligned processes, and secure communication protocols to safeguard sensitive information throughout the audit lifecycle.
Contact us
Whether you need a full GCP audit programme or targeted support for a specific study, vendor, or inspection, our experts can help you strengthen compliance and inspection readiness.
If you’re interested in our consultancy services, find out more about our QA services and get in touch today
Helpful resources
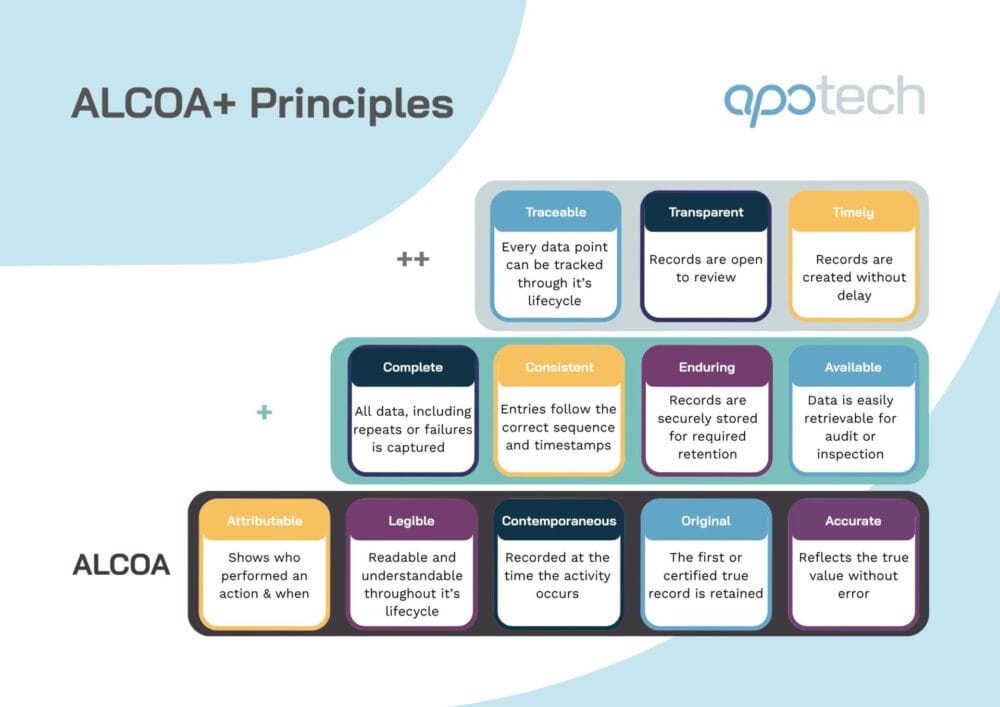
ALCOA+ Principles in Pharma: The Next-Gen Standard for Data Integrity
ALCOA+ and the modern data integrity landscape Data integrity underpins regulatory compliance and patient safety. And as pharmaceutical and biotech organizations adopt increasingly digital systems, automated workflows, and globalized supply chains, regulators including the FDA, EMA, and MHRA have intensified their focus on how data is generated, captured, and maintained. The ALCOA+ principles form the…
GxP Compliance Explained: Beyond Best Practice
Understanding the meaning of GxP is essential for any organization operating in the life sciences sector. In simple terms, GxP refers to “Good Practice” quality and regulatory guidelines, used to ensure products are safe, effective, and reliable throughout their lifecycle. These standards apply across pharmaceuticals, biotechnology, medical devices, and laboratory environments, and are enforced by…
How To Master The CAPA Process Step-by-Step: From Identification to Implementation
The Corrective and Preventive Action (CAPA) process is a core mechanism within Pharmaceutical and Biotech quality systems, designed to identify, address, and prevent issues that may impact compliance, product quality, and most importantly, patient safety. In regulated environments, CAPAs are not isolated actions but structured responses embedded within a wider quality management framework. CAPAs are commonly triggered by internal…